Naetur is a brand of 100% organic plant-based dietary supplements that promote better sleep, improved digestion, reduced fatigue, and enhanced recovery.

Can the microbiota help manage stress and anxiety?
Can the microbiota help manage stress and anxiety?
In this article, you will discover what stress is, how it affects your body, and how to manage it effectively. Nahibu, as a specialist in gut microbiota, has decided to include a module on the role of gut bacteria in stress and anxiety resistance in its microbiota analysis. You will therefore discover how bacteria in your gut can regulate your brain!

What is stress?
Nowadays, few people can claim that they are not stressed. Our societies often impose an ideal of social, professional, and family success that can cause a lot of pressure. Juggling different activities, your professional life, and finding time for yourself can be difficult. Many people say they are stressed. What exactly does that mean?
Stress is a complex phenomenon that triggers emotional, biological, and physical responses. The response to stress is different for each person depending on genetics, environment, and diet.
Stress can be broken down into a trigger (the stressor), a compensatory reaction, and the establishment of a new balance.
There is acute stress, which does not last long, and chronic stress, which lasts over time and can be harmful.
Anxiety, on the other hand, is the tendency to anticipate or dwell on difficulties, thereby amplifying them. It is therefore a source of stress.
Anguish is a specific but intense psychological experience, characterized by a feeling of loss of control and the imminence of serious danger. Anguish manifests itself through several symptoms, including a feeling of oppression, breathing difficulties, and a rapid heartbeat. It is linked to anxiety, which is a more chronic and less destabilizing condition. Anxiety and nervousness are two conditions that cause stress.
What are the triggers and symptoms of stress?
The factors responsible for stress, or stressors, can be internal or external, real or imagined, such as an unpleasant person in one's circle, an illness, or even a car breakdown. Stressors trigger a physiological crisis mode response in the body, the purpose of which is to adapt and restore balance.
The symptoms of stress can be very diverse and include fatigue, sleep disorders, loss of appetite or overeating, skin conditions such as eczema, irritability, decreased libido, heart palpitations, and muscle tension.
The symptoms of an anxiety attack, which is a short and very intense form of anxiety, include heart palpitations, sweating, chest pain, tremors, a feeling of suffocation, dizziness, nausea, and stomach pain. They are accompanied by psychological symptoms such as the feeling of going crazy or imminent death. These symptoms are very frightening and themselves fuel the panic or anxiety attack.
Anxiety is a state of distress that is less intense but more long-lasting. Some symptoms of an anxiety attack may be present, but they are less intense, such as nausea or stomach ache and palpitations.
How important is stress?
The number of people affected by stress and anxiety is constantly growing, mainly due to our lifestyles and socio-economic models. In addition, the health crisis triggered by the emergence of the SARS-CoV-2 virus in late 2019 has contributed to an increase in these disorders worldwide. It has changed our habits, led to the isolation of a large part of the population, called into question many certainties, and increased mortality rates in many countries.
The prevalence of anxiety disorders was estimated at 21.6% in 2010. Women, young people, and low-income individuals were the most at risk. Depressive episodes, alcohol abuse, and drug addiction are often associated with anxiety disorders.
According to a 2012 study, 22% of European workers experience stress.
The COVID-19 health crisis has caused a dramatic increase in stress and anxiety levels: the prevalence of stress is currently estimated at 29.6% and that of anxiety at 31.9%.

What happens in the body under the effects of stress?
The stress response refers to the changes that take place in the body, such as behavioral changes or the secretion of specific hormones. Stress has negative effects if it is severe or prolonged, i.e., if it becomes chronic.
Acute stress occurs when the body is subjected to a threat such as an accident, illness, or a stressful psychological situation that does not last long. Reactions are then triggered to either fight or flee (fight or flight). This is a legacy from our ancestors millions of years ago. When faced with a dangerous animal such as a lion, the threat was perceived by the body, which adapted to increase the chances of survival: flee from the animal or fight it. Nowadays, it is rare to encounter a wild animal, but this mechanism of response to a threat is still present. The physiological changes associated with the stress response are:
- The mobilization of energy to maintain brain and muscle functions
- Focusing attention on the perceived threat
- Increased glucose utilization
- Increased blood flow and respiration, redistribution of blood flow, increased energy supply to muscles and brain
- Modulation of immunity
- Inhibition of reproductive physiology and sexual behavior
- Decreased hunger and food intake
The brain perceives the threat and signals the adrenal glands (located above the kidneys) to release sufficient amounts of adrenaline. This increases blood and oxygen flow to the muscles, allowing them to perform optimally, and increases brain alertness.
If acute stress persists, cortisol, also known as the stress hormone, is produced by the adrenal glands to increase blood glucose levels, which provide energy to the brain and muscles.
During chronic stress, cortisol is produced continuously. This can have negative effects on the body, as this state leads to excessive fatigue, reduced immunity, disturbed sleep, metabolic problems, and dietary changes that can cause weight loss or gain. Chronic stress thus depletes energy reserves and alters neurotransmitter production. Neurotransmitters are molecules that send signals to neurons, such as serotonin, which regulates many mental and behavioral processes, and melatonin (nicknamed the sleep hormone).
What is the difference between stress, anxiety, anguish, and fear?
Fear, anxiety, and distress or panic are different but related psychological manifestations. Fear can cause anxiety, distress is a very intense form of anxiety, and anxiety can cause stress.
Fear is a disproportionate aversion to something that is characterized by tension, trembling, or sweating.
Anxiety is a tendency to anticipate or dwell on negative things, which can cause stress.
An anxiety attack or panic attack is an intense experience closely linked to anxiety, with very dramatic symptoms such as a feeling of losing control.
Is there a treatment for stress?
There is no miracle cure for stress and anxiety, but certain sophrology or meditation techniques can help you accept your emotions and manage how you react to them.
Exercise and a balanced diet can also help support your body. One study also showed that listening to relaxing music helped lower salivary cortisol levels more quickly after acute stress.
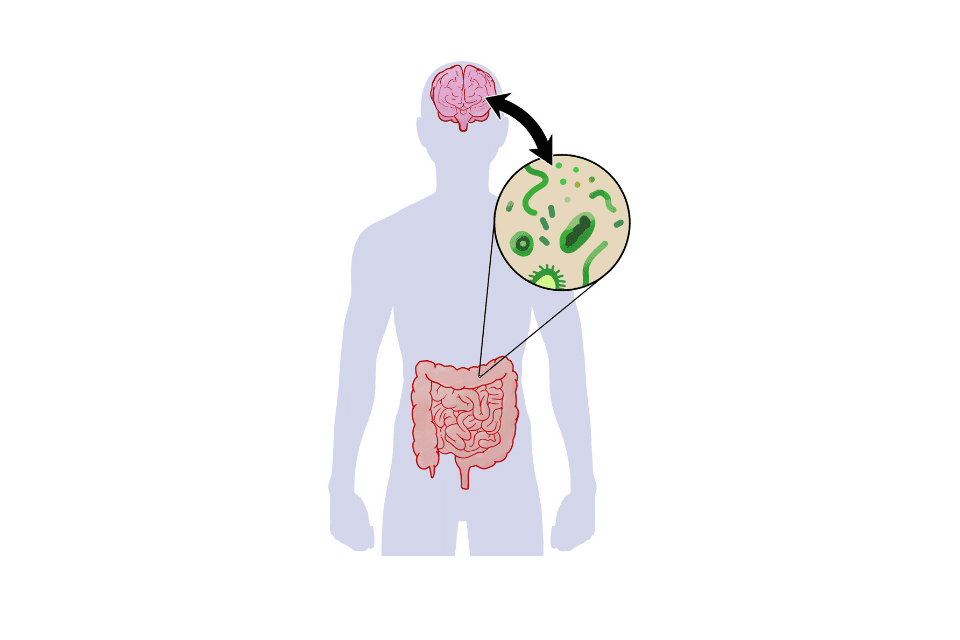
How do the gut and microbiota regulate our stress?
Within the enteric nervous system, there are one to several hundred thousand neurons that participate in the gut-brain axis. This communication axis allows for constant, two-way dialogue between the two organs. One of the most concrete manifestations of the existence of the gut-brain axis is the alteration of transit (diarrhea or constipation) caused by a stressful event such as public speaking or an exam.
The gut-brain axis is regulated by the gut microbiota, the collection of microorganisms living in the lower part of our digestive tract. Certain microbes, such as intestinal bacteria, produce signals that modulate the messages sent to the brain, known as neurotransmitters. Serotonin, for example, also known as the happiness hormone, is produced largely by the gut microbiota. In addition to our mood and behavior, it also regulates the intestinal barrier. Its production from the amino acid tryptophan is impaired in cases of sustained stress.
Finally, stress can also damage the intestinal barrier that protects our body from bacterial or food residues passing into our cells. If this barrier is impaired, low-level inflammation can develop in the intestine.
All of this tends to demonstrate that there is an important link between the microbiota, the intestinal barrier, and stress and anxiety.

How can we measure the impact of our gut microbiota on stress and anxiety?
Studies have shown that gut microbiota regulates stress response and anxiety levels. Initial observations have also concluded that taking certain probiotics consisting of Lactobacillus and Bifidobacteria can reduce stress and anxiety. However, many claims about the effect of probiotics on our mental health still lack clinical evidence.
How can you find out if your microbiota could potentially help you resist stress and anxiety? Take the test and find out in your Nahibu results!
What foods should you eat to combat stress and anxiety?
What should you eat to manage stress? You can increase your serotonin levels by eating a diet rich in complex sugars (whole grains and plant-based products) and low in protein. Tryptophan, from which serotonin is formed, is found in whole grains, but also in chocolate.
Omega-3s are fats that are beneficial for the brain and are associated with a reduced risk of depression. They are found in oily fish, nuts such as walnuts, hazelnuts, almonds, and cashews, and rapeseed, flaxseed, and walnut oils. Feel free to replace meat with fish and use healthy oils to dress your salads.
Plant-based foods such as fruits, vegetables, and legumes contain vitamins that help the body function properly and contribute to good stress resistance.
Stress can cause a decrease in magnesium levels, which in turn can exacerbate its effects. A diet rich in magnesium can therefore help combat stress and anxiety. So don't hesitate to eat dark chocolate made with pure cocoa butter in reasonable quantities, oilseeds, and seafood to optimize your magnesium intake. You can discover more foods to improve your resistance to stress and anxiety thanks to Shido's personalized nutrition algorithm.
How can you manage your stress?
A healthy lifestyle (regular bedtimes, little or no alcohol, and daily physical activity) and a balanced diet can help reduce stress and anxiety levels. In addition, certain foods will be your best allies, such as plant-based products, fatty fish (salmon, herring, tuna, mackerel, etc.), oilseeds, and dark chocolate. Why not replace sugary foods such as cookies or other dessert creams with a handful of almonds or walnuts and a square of dark chocolate?
When faced with difficult emotions, why not try mindfulness meditation or sophrology techniques, which are very effective in reducing stress? They will also teach you not to fight stress but to accept it in order to manage it better. Give yourself time for yourself and enjoy the present moment, alone or with your loved ones. Don't be ashamed to discuss your stress and anxiety with those around you, and seek professional help if necessary.
Finally, test your gut microbiota with Nahibu to discover how your gut flora may contribute to stress and anxiety management, and get personalized dietary advice on how to improve it!
Sources:
Prevalence, characteristics and comorbidities of anxiety disorders in France: Results from the Mental Health in General Population Survey (MHGP). E. Leray, A. Camara, D. Drapier, F. Riou, N. Bougeant, A. Pelissolo, K.R. Lloyd, V. Bellamy, J.L. Roelandt, B. Millet. European Psychiatry, 2011.
Prevalence of stress, anxiety, depression among the general population during the COVID-19 pandemic: a systematic review and meta-analysis. Nader Salari, Amin Hosseinian-Far, Rostam Jalali, Aliakbar Vaisi-Raygani, Shna Rasoulpoor, Masoud Mohammadi, Shabnam Rasoulpoor and Behnam Khaledi-Paveh. Globalization and Health, 2020.
Effects of relaxing music on salivary cortisol level after psychological stress. Stéphanie Khalfa, Simone Dalla Bella, Mathieur Roy, Isabelle Peretz, Sonia J. Lupien. Ann. New York Academy of Sciences, 2003.
Breaking down the barriers: the gut microbiome, intestinal permeability and stress-related psychiatric disorders. John R. Kelly, Paul J. Kennedy, John F. Cryan, Timothy G. Dinan, Gerard Clarke, and Niall P. Hyland. Frontiers in Cellular Neuroscience, 2015.
Gut/brain axis and the microbiota. Emeran A. Mayer, Kirsten Tillisch, and Arpana Gupta. The Journal of Clinical Investigation, 2015.
Nutrient and Stress Management, Karuna Singh. The Journal of Nutrition and Food Sciences, 2016.
Discover more articles on the microbiota.
Partnership between Naetur and Nahibu
Partnership between the EffiNov Laboratory and Nahibu
The French laboratory EffiNov, which specializes in micronutrition, develops a range of dietary supplements and functional foods. Their products are formulated by a team with multidisciplinary expertise (medical, nutritional, and scientific).
Partnership between Umamiz and Nahibu
Umamiz offers products made from spirulina, a cyanobacterium with impressive properties. Whether it's Spirulina Givrée or Véritable Spirulina, the spirulina is 100% French and produced on the Umamiz farm.
Take care of your microbiota with Nahibu.