Salvia specializes in aromatherapy, particularly in the formulation of essential oil blends. The advantages: controlled dosages that would be impossible to reproduce at home, careful study of each raw material and monitoring throughout the manufacturing process, safety for all users, and optimal effectiveness.

Acting on short-chain fatty acid production
Acting on your production of short-chain fatty acids
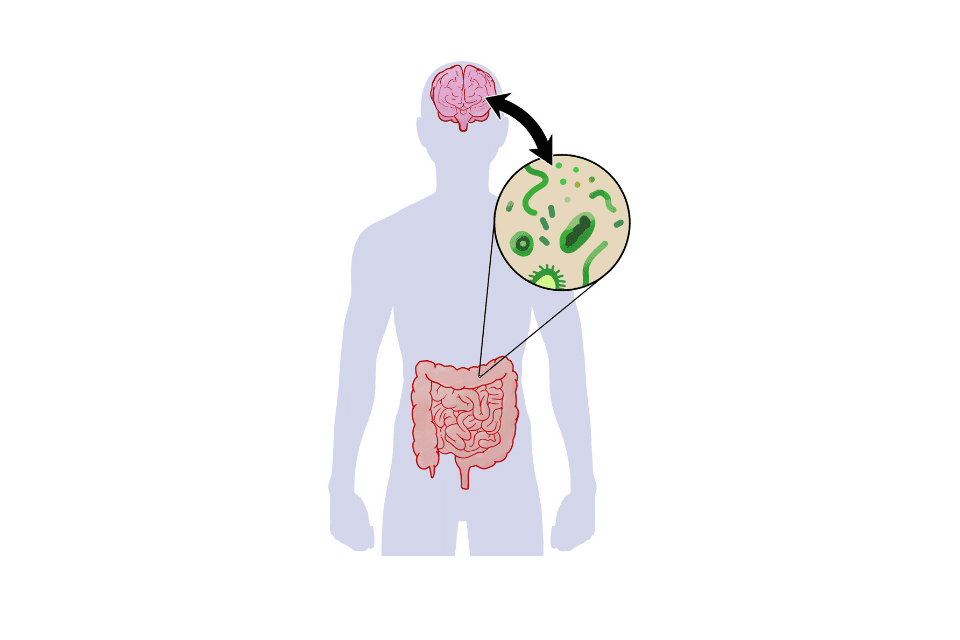
The gut microbiota
Our health ally
The body evolves in symbiosis with the ecosystem it harbors in the digestive tract. It is composed of a multitude of microorganisms (viruses, bacteria, archaea, fungi) and forms the gut microbiota. Each of these bacteria plays a key role in many vital functions of the body and also in maintaining good health. However, if the balance is disrupted, bacteria that were initially beneficial to health can become harmful and lead to disorders such as neurodegenerative diseases (Alzheimer's, Parkinson's, etc.), metabolic diseases (diabetes, obesity), cancers, and allergies.
Factors that alter the gut microbiota
The gut microbiota develops from birth until around the age of two and a half. After that, it is considered stable and its composition will only change due to certain factors such as illness, stress, medication, health, diet, geographical location, etc. Under the influence of these factors, the bacterial composition may change.
How can we influence the gut microbiota?
Today, a link has been established between diet and the bacterial composition of the gut microbiota. It is therefore important to eat a varied and balanced diet in order to optimize your microbiota and enjoy its benefits. For example, it is recommended to increase your intake of dietary fiber at the expense of simple carbohydrates. In addition to being filling and beneficial for intestinal transit, fiber reduces the risk of diabetes, cancer, and cardiovascular disease. Finally, it also nourishes good intestinal bacteria.

Carbohydrates, more commonly known as sugars, are molecules whose function is to provide energy to the body in the form of calories. There are two types of carbohydrates: simple (or monosaccharides) and complex (or polysaccharides). Simple carbohydrates such as glucose, fructose, and galactose are non-hydrolyzable molecules, meaning they cannot be broken down into smaller molecules.
Complex carbohydrates, on the other hand, are formed by the linking of simple carbohydrates. These include disaccharides such as lactose and sucrose, which are composed of two simple carbohydrates, and polysaccharides (starch, cellulose, glycogen, etc.), which are composed of more than two simple carbohydrates. Only simple carbohydrates and disaccharides in complex carbohydrates have sweetening power. Polysaccharides do not.

Dietary fiber is a complex carbohydrate of plant origin that is neither digested nor absorbed by the body. There are two types of fiber: soluble fiber and insoluble fiber. Fiber is naturally present in plant-based foods, but its distribution varies. Vegetables (artichokes, beets, carrots, broccoli, cabbage, etc.), legumes (beans, lentils, chickpeas, etc.), whole grains (oats, spelt, brown rice, etc.), fruits (apples, pears, oranges, grapefruit, etc.) and dried fruits (dates, prunes, etc.) are excellent sources.
Digestion of dietary fiber
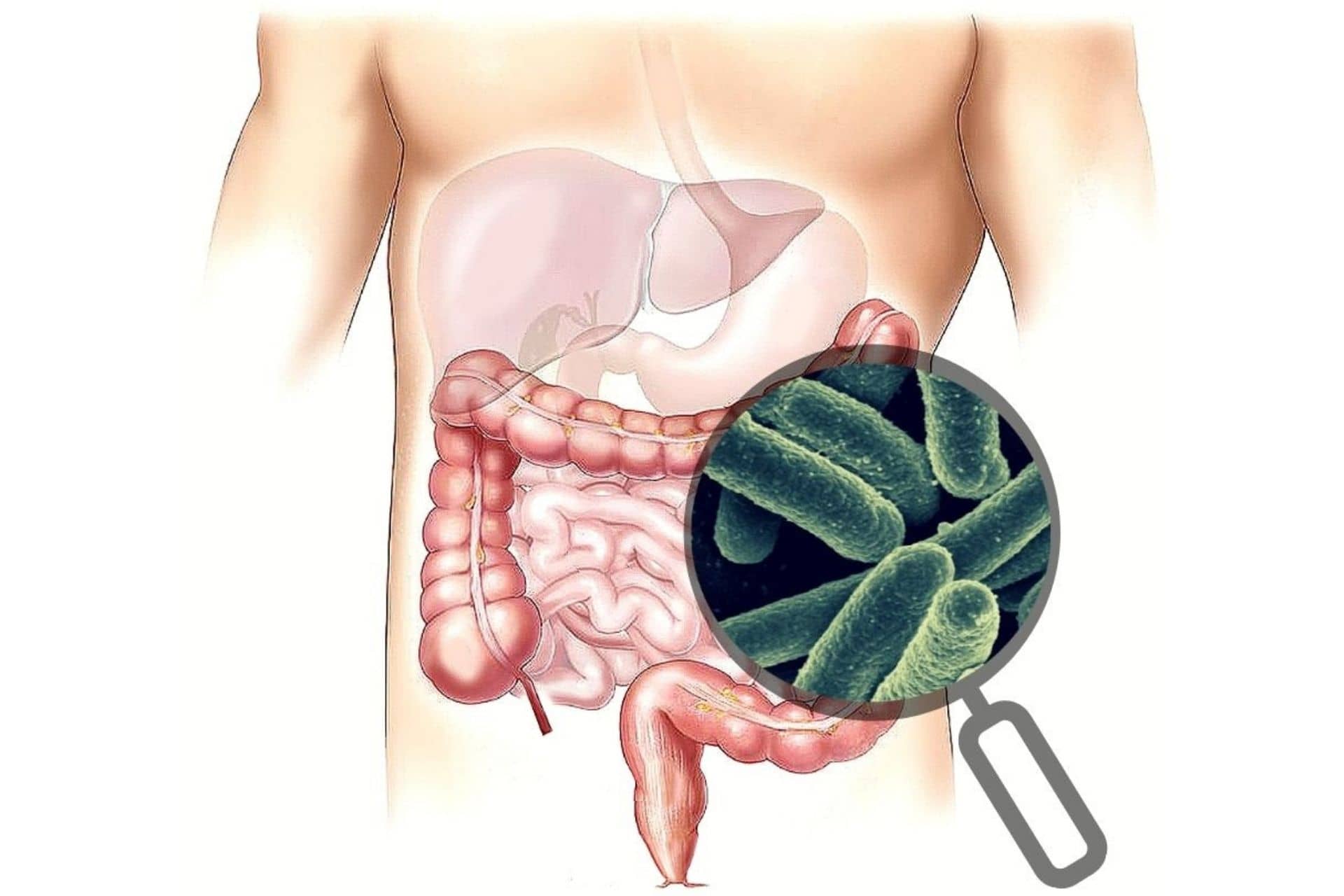
Once ingested, dietary fiber passes through the digestive tract to the colon without being digested or absorbed. It is in the colon that it first undergoes hydrolysis (or dissociation) by enzymes and then fermentation by bacteria, releasing short-chain fatty acids. Some of these acids perform their functions in the intestine, while others migrate and act throughout the body.

Short-chain fatty acids
What are SCFAs?
Short-chain fatty acids (SCFAs) are a subset of fatty acids synthesized by the gut microbiota during the fermentation of non-digestible polysaccharides such as dietary fiber. Put simply, they are molecules produced by good gut bacteria during the fermentation of food. There are several types, such as acetate, butyrate, lactate, propionate, and succinate, which vary in proportion and function. Acetate, butyrate, and propionate are the most common. Each plays an important role in maintaining health and in the development of disease.
Where do SCFAs act?
Once produced, SCFAs can remain in the colon and be used by intestinal cells or be transported into the bloodstream to act on other organs. The production of SCFAs is influenced by various factors. It depends on the number of bacterial species present in the gut microbiota. The source of dietary fiber and transit time also play a role.
Take care of your microbiota with Nahibu.

What are the health effects of SCFAs?
AGCCs are now recognized as mediators of the beneficial effects of dietary fiber and gut microbiota on host health. First, they play a key role in maintaining intestinal health. They improve the integrity of the intestinal barrier by regulating pH and mucus production and providing the energy necessary for epithelial cells to perform their functions. They also modulate inflammatory and immune responses. They regulate immune cell function and lymphocyte differentiation. Lymphocytes are white blood cells that are part of the immune system and protect the body against attacks (bacteria, viruses, diseases, etc.). They also limit the proliferation of tumor cells. In addition, they act on skeletal muscles by modulating their function and exercise capacity. Finally, they prevent the risk of gastrointestinal disorders, hypertension, insulin resistance, obesity, cancer, and cardiovascular disease.
How can you find out your AGCC level?
It is now possible to analyze your gut microbiota. In addition to detecting the depletion of bacterial biodiversity associated with the risk of dysbiosis, the analysis also detects short-chain fatty acids.
How can you increase your SCFA levels?
As diet is the main factor influencing the amount of short-chain fatty acids, it is now essential to include fiber-rich foods in your diet to stimulate their production. The higher your fiber intake, the more SCFAs will be produced.

Focus on butyrate
A fiber-rich diet is extremely beneficial to health. Increasing butyrate levels has several benefits. In addition to being the main source of energy for epithelial cells, it plays a key role in preventing cancer and inflammatory bowel disease. Furthermore, butyrate supplementation has been shown to reduce the risk of obesity.
Focus on succinate
Succinate, on the other hand, appears to have negative effects. High levels of succinate are found in the intestinal lumen in patients with dysbiosis or inflammatory bowel disease. However, its involvement remains to be confirmed.
In conclusion, it is essential to ensure sufficient dietary fiber intake to enable the microbiota to produce short-chain fatty acids and thus promote good health. In addition to their benefits for the microbiota, fibers are essential for the body. They act as our allies for optimal well-being and to limit the risk of developing certain diseases. Short-chain fatty acids are considered key mediators between diet, gut microbiota, and health. Nahibu, a player in the food industry of tomorrow, offers a gut microbiota analysis solution that measures its diversity and detects SCFAs.
Sources:
Jian Tan, Craig McKenzie, Maria Potamitis, Alison N Thorburn, Charles R Mackay, Laurence Macia. The role of short-chain fatty acids in health and disease. 2014.
Sean M McNabney, Tara M Henagan. Short Chain Fatty Acids in the Colon and Peripheral Tissues: A Focus on Butyrate, Colon Cancer, Obesity and Insulin Resistance. 2014.
Yao Yao, Xiaoyu Cai, Weidong Fei, Yiqing Ye, Mengdan Zhao, Caihong Zheng. The role of short-chain fatty acids in immunity, inflammation and metabolism. 2020.
James Frampton, Kevin G Murphy, Gary Frost, Edward S Chambers. Short-chain fatty acids as potential regulators of skeletal muscle metabolism and function. 2020.
E E Blaak, E E Canfora, S Theis, G Frost, A K Groen, G Mithieux, A Nauta, K Scott, B Stahl, J van Harsselaar, R van Tol, E E Vaughan, K Verbeke. Short chain fatty acids in human gut and metabolic health. 2020.
Julia M W Wong, Russell de Souza, Cyril W C Kendall, Azadeh Emam, David J A Jenkins. Colonic health; fermentation and short chain fatty acids. 2006.
Sathish Sivaprakasam, Puttur D Prasad, Nagendra Singh. Benefits of short-chain fatty acids and their receptors in inflammation and carcinogenesis. 2016.
Discover more articles on the microbiota.
Partnership between Salvia and Nahibu
Partnership between the Inolab laboratory and Nahibu
Specializing in micronutrition, Inolab produces innovative, additive-free dietary supplements inspired by nature.
Partnership between Uberti and Nahibu
Experts in nutrition and naturopathy, Uberti develops 100% organic and natural products for a healthier and more balanced diet.
Take care of your microbiota with Nahibu.